 For example, let’s calculate the mass of 2 moles of table salt, sodium chloride or NaCl. Thus the mass m of substance in grams is equal to the quantity n in moles multiplied by the molar mass M of the substance in g/mol. The formula used to convert moles to grams is: m n × M. I put together this quick reference guide (below) to help my students. Step Two: Use the Moles to Grams Formula. Use the periodic table to determine the molar mass of AgCl, which is 143.321 g/mol.Ĭheck out the above video if you want more details for each step. Convert moles of B to grams of B (moles of AgCl to grams of AgCl).This is to cancel out BaCl 2 and leave AgCl. How many grams are in 3. Its actually one of seven base units of the International System of Units. Moles to Grams Conversion Formula Questions: 1. n m / M What is a mole A mole simply refers to a unit for measurement. Note the moles of BaCl 2 are on the bottom. To accurately calculate the number of moles n in a substance of a certain mass m (in grams), you must use the grams-to-moles formula. There’s a 1 mole BaCl 2 to 2 mole AgCl ratio. Use the coefficients in the balanced chemical equation to find the molar ratio. Convert moles of A to moles of B (moles of BaCl 2 to moles of AgCl).Step 4: Then continue conversions until you reach the units and substance asked for. This is to cancel out what we are given (grams), and convert to moles. Notice that the molar mass is placed with the grams at the bottom and mole at top. The molar mass for BaCl 2 is 208.233 g/mol. And we determine this using the periodic table. So, we need to convert grams of BaCl 2 to moles of BaCl 2. Step 3: Make sure your given is in moles – If grams/liters/particles of A, convert the moles of A Step 2: Set up the multiplication grid – Put the given on the ‘front ledge’, and what you want to find behind the grid. We are given 48.5 g of BaCl 2, and being asked to find grams of AgCl. Step 2: Determine the given and what is being asked Step 1: Always have a balanced chemical equation! Problem: If 48.5 g of barium chloride reacts in excess with silver nitrate, how many grams of silver chloride are produced? Now, let’s use the steps to solve a problem. It walks through each conversion with more detail. Want more info for each step? Here’s a video on mole to mole, mole to grams, and grams to grams conversion. PRO TIP: Substance A must be in moles before converting to Substance B! Then continue conversions until you reach the units and substance asked for.Make sure your given is in moles – If grams/liters/particles of A, convert the moles of A.Set up the multiplication grid – Put the given on the ‘front ledge’, and what you want to find behind the grid. 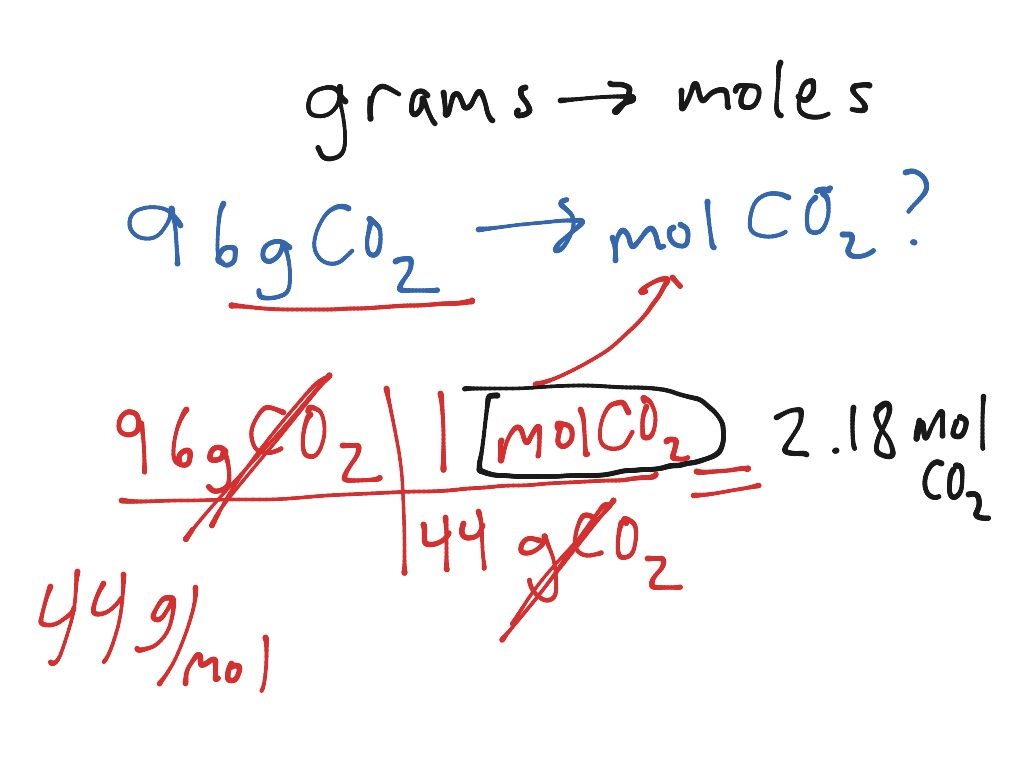
Determine the given and what is being asked.Always have a balanced chemical equation!.And since tracking each unit conversion is so important, always use a multiplication grid! That’s the T-chart looking thing that your teacher is always asking you to use. Well…actually the key to success is keeping up with the units. 3 Moles of Oxygen Note: 1 Mole of Oxygen 15.9994 grams (about 13.5 Cubic Feet) From the table above, we can see that 1 Mole of Silver Nitrate weighs. The challenge is keeping up with the units. Units (moles, grams, liters, particles).So, you may be converting both the Substance (NaCl, Fe, CO 2) For example, you may be given moles of A and need to convert to grams of B. We use these relationships to convert from one substance/unit to another. Specifically, between the reactants and products in chemical reactions. (Hint: Convert grams to moles before attempting to answer the question).Stoichiometry is all about relationships. Is there enough H 2 to react with the O 2 you have? You have 64.00 g of O 2 and 2.016 g of H 2. In order to react fully without leaving anything leftover every mole of oxygen gas ( O 2) must react with two moles of hydrogen gas ( H 2):.How many moles of oxygen atoms do you have for each of the following? They perform the same function as counting how many items you have. For example, one molecule of H 2O has two atoms of H and 10 molecules of H 2O has 20 atoms of H. You can count atoms within molecules, too.Why didn’t you get the same number of moles? 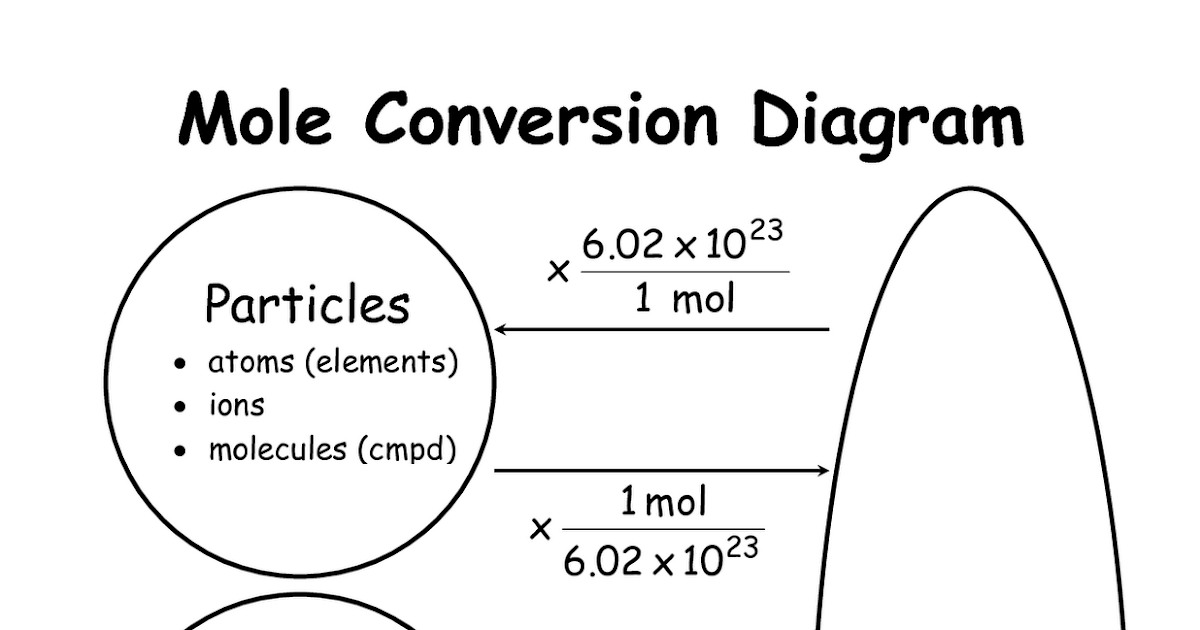
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
